This is approximately the sum of the number of protons and neutrons in the nucleus. The mass of an atom relative to that of carbon-12. The transition of a substance directly from the solid to the gas phase without passing through a liquid phase.ĭensity is the mass of a substance that would fill 1 cm 3 at room temperature. The temperature at which the liquid–gas phase change occurs. The temperature at which the solid–liquid phase change occurs. The arrangements of electrons above the last (closed shell) noble gas. These blocks are named for the characteristic spectra they produce: sharp (s), principal (p), diffuse (d), and fundamental (f). 
The atomic number of each element increases by one, reading from left to right.Įlements are organised into blocks by the orbital type in which the outer electrons are found. Members of a group typically have similar properties and electron configurations in their outer shell.Ī horizontal row in the periodic table. In the end, for this particular geometry, it doesn't matter whether you assume the bottom four #d# orbitals are in a high spin or low spin configuration amongst each other.A vertical column in the periodic table. So, we would expect to fill the lower four #d# orbitals completely, before filling the #d_(z^2)#. Here we see that the energy gap between #d_(xy)# and #d_(x^2 - y^2)# and #d_(z^2)# is large. The crystal field splitting diagram then looks like this: The #d_(xz)# and #d_(yz)# are about equally stabilized amongst each other, but more stabilized than the #d_(xy)# and #d_(x^2-y^2)#, as the best the ligands can do is line up with their nodal planes.The #d_(xy)# and #d_(x^2 - y^2)# are somewhat stabilized by their attraction to the positive metal center.The #d_(z^2)# is directly along the #z# axis, and is highly destabilized by interacting directly with the metal #d# orbital. 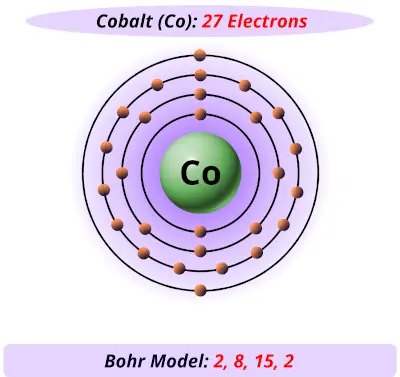
Here, we treat the #"CN"^(-)# as point charges that repel the metal's #d# orbitals as they come in to form the complex. I won't go too much into the molecular orbital diagram other than the portion of it that comes out of Crystal Field Theory. #underbrace(ul(uarr darr)" "ul(uarr color(white)(darr))" "ul(uarr color(white)(darr))" "ul(uarr color(white)(darr))" "ul(uarr color(white)(darr)))_("3d")#īut this is a complex, and cobalt here has a coordination number of #5#, which denotes a trigonal bipyramidal geometry. So the electron configuration for #"Co"^(3+)# isĪnd we denote complex as containing a #bb(d^6)# electron count. The electron configuration of neutral cobalt is Thus, cobalt is in its #bb(+3)# oxidation state. Since cyanide, #"CN"^(-)# will contribute a #-1# charge, it follows that the total charge contributed by #5# of them is #-5#. If there are any unpaired electrons, we should expect #^(2-)# to be paramagnetic. (We'll work in the realm of crystal field theory and only look at the crystal field splitting diagram that came from the metal's original #d# atomic orbitals.) Well, the first thing you should do is find out the oxidation state of cobalt in this complex ion, so you can find its d-electron count.įrom there, by knowing the electron configuration of cobalt in the complex, one can deduce if any of the electrons are unpaired in the complex's molecular orbital diagram. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
